Atomic Structure, atomic number and mass number
A couple of revision videos if you prefer animation to static text. You require a slightly longer attention span than that of a goldfish for each video.
A couple of revision videos if you prefer animation to static text. You require a slightly longer attention span than that of a goldfish for each video.
In the first of three posts about naming and drawing structures for organic molecules we classified hydrocarbons as either saturated or unsaturated. We learned how to systematically name alkanes, a family of hydrocarbons with single covalent bonds only,
There are three main types of chemical formula in organic chemistry each providing different information. The formula used will depend on the circumstances
Salbutamol. the molecule on the right represents yet another type of formula you will come across regularly. Which of the three formula types listed is it most closely related to and why? It can save a lot of space if you miss out all the carbon atoms and just show the covalent bonds in the skeleton. The carbon atoms sit in the corners. If only three covalent bonds come to a point then you know that there is a hydrogen atom attached at that point as qwell.
Look at the structure carefully and work out the molecular formula for salbutamol. Check your answer by going to chemspider and plug salbutamol into the search tool. This is a useful site if you find the name of an ingredient on a product and want to find out more about it. For instance triclosan is a molecule receiving a lot of attention at the moment. Check it out on chemspider and find out why it is a rather controversial molecule.
Finally you need to be competent moving from a full structural formula to a condensed structural formula and vice versa. Make sure that you have got the hang of this by watching the short 4 minute video
You can trial the full powerpoint here. The resource is fully animated and scaffolded. It can be used in the classroom for teacher led instruction. It could be used as a student self learning programme in a computer room or for homework.
You can purchase the powerpoint and associated assessment exercises on my online store at teachers pay teachers

 Organic chemistry is undoubtedly the most important branch of chemistry. At the molecular level it can be the study of life itself. A washing machine sized robot named Philae has just started an eternal piggy back ride on a lump of rock dating back to the beginning of time. Its mission to discover if life starting molecules are part of the frozen package. A billion dollar launch and ten years later a permanent stop off on an asteriod where it will drill below the surface sniffing out organic compounds before drifting through the cosmos till the end of time. Organic chemistry started the moment supernovae starting spewing carbon atoms into the universe. Its an important subject Ok got a bit carried away. Organic chemistry is the study of carbon and its compounds with hydrogen and a few other elements mainly oxygen nitrogen phosphorus and sulphur
Organic chemistry is undoubtedly the most important branch of chemistry. At the molecular level it can be the study of life itself. A washing machine sized robot named Philae has just started an eternal piggy back ride on a lump of rock dating back to the beginning of time. Its mission to discover if life starting molecules are part of the frozen package. A billion dollar launch and ten years later a permanent stop off on an asteriod where it will drill below the surface sniffing out organic compounds before drifting through the cosmos till the end of time. Organic chemistry started the moment supernovae starting spewing carbon atoms into the universe. Its an important subject Ok got a bit carried away. Organic chemistry is the study of carbon and its compounds with hydrogen and a few other elements mainly oxygen nitrogen phosphorus and sulphur
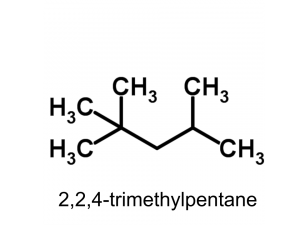 This is a simple molecule but even governments of small countries are prepared to go war to secure supplies of this hydrocarbon. Its a hydrocarbon because it contains the elements carbon and hydrogen only. The research octane number or RON of gasoline is a measure of its efficiencey compared with 2,2,4-trimethyl pentane under test conditions. The number is displayed at the pump. Just to confuse you this molecule also goes by the name isooctane.
This is a simple molecule but even governments of small countries are prepared to go war to secure supplies of this hydrocarbon. Its a hydrocarbon because it contains the elements carbon and hydrogen only. The research octane number or RON of gasoline is a measure of its efficiencey compared with 2,2,4-trimethyl pentane under test conditions. The number is displayed at the pump. Just to confuse you this molecule also goes by the name isooctane.
How do we write structural formulae for organic molecules and give them a unique name that everyone understands (Chemists call this a systematic name). I have tried to explain this in the video starting with the simplest organic molecules, the hydrocarbons. Back to the cosmos again The fuel that carried Rosetta away from earth on a trajectory that would see it 10 years later bounce along the surface of a comet was a hydrocarbon, hydroxyl terminated polybutadiene (HTBD). On this planet the vast majority of polybutadiene is used in tyre manufacture for vehicles driving on high octane fuel
Stop the video when prompted and attempt the questions towards the end to check understanding.
You can purchase the powerpoint and associated assessment exercises on my online store at teachers pay teachers

Trial the powerpoint first
Work through The Introduction to the Periodic Table Prezi then check your understanding using the worksheet
By the end of this unit you should know:
what the atomic number and mass number of an element is.
how elements are arranged on the periodic table.
what Periods and Groups are.
the physical and chemical properties of the group 1 metals.
the physical properties of group 17 elements.
the physical properties of group 18 elements
that sodium and chlorine react violently together.
that elements in the same group have similar properties. WHY!
 Work through the first 14 slides of the presentation then do the short test at the end of this post.
Work through the first 14 slides of the presentation then do the short test at the end of this post.
 AS 90937 NCEA Level 1 Electricity and Magnetism covers the following topics
AS 90937 NCEA Level 1 Electricity and Magnetism covers the following topicsAS