What have we done so far?
In the first of three posts about naming and drawing structures for organic molecules we classified hydrocarbons as either saturated or unsaturated. We learned how to systematically name alkanes, a family of hydrocarbons with single covalent bonds only,
Formulae in organic chemistry, how much information?
There are three main types of chemical formula in organic chemistry each providing different information. The formula used will depend on the circumstances
- Molecular Formula: The molecular formula shows the numbers and types of atoms in the molecule
- Full Structural Formula: A full structural formula shows all the covalent bonds in the molecule.
- Condensed structural formula: A condensed structural formula shows carbon atoms with the total number of atoms bonded with them.
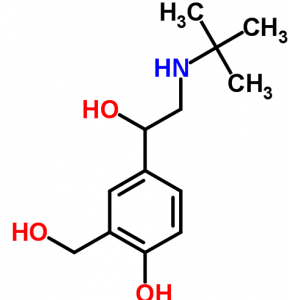
Salbutamol. the molecule on the right represents yet another type of formula you will come across regularly. Which of the three formula types listed is it most closely related to and why? It can save a lot of space if you miss out all the carbon atoms and just show the covalent bonds in the skeleton. The carbon atoms sit in the corners. If only three covalent bonds come to a point then you know that there is a hydrogen atom attached at that point as qwell.
Look at the structure carefully and work out the molecular formula for salbutamol. Check your answer by going to chemspider and plug salbutamol into the search tool. This is a useful site if you find the name of an ingredient on a product and want to find out more about it. For instance triclosan is a molecule receiving a lot of attention at the moment. Check it out on chemspider and find out why it is a rather controversial molecule.
Finally you need to be competent moving from a full structural formula to a condensed structural formula and vice versa. Make sure that you have got the hang of this by watching the short 4 minute video
For Educators
You can trial the full powerpoint here. The resource is fully animated and scaffolded. It can be used in the classroom for teacher led instruction. It could be used as a student self learning programme in a computer room or for homework.
You can purchase the powerpoint and associated assessment exercises on my online store at teachers pay teachers